 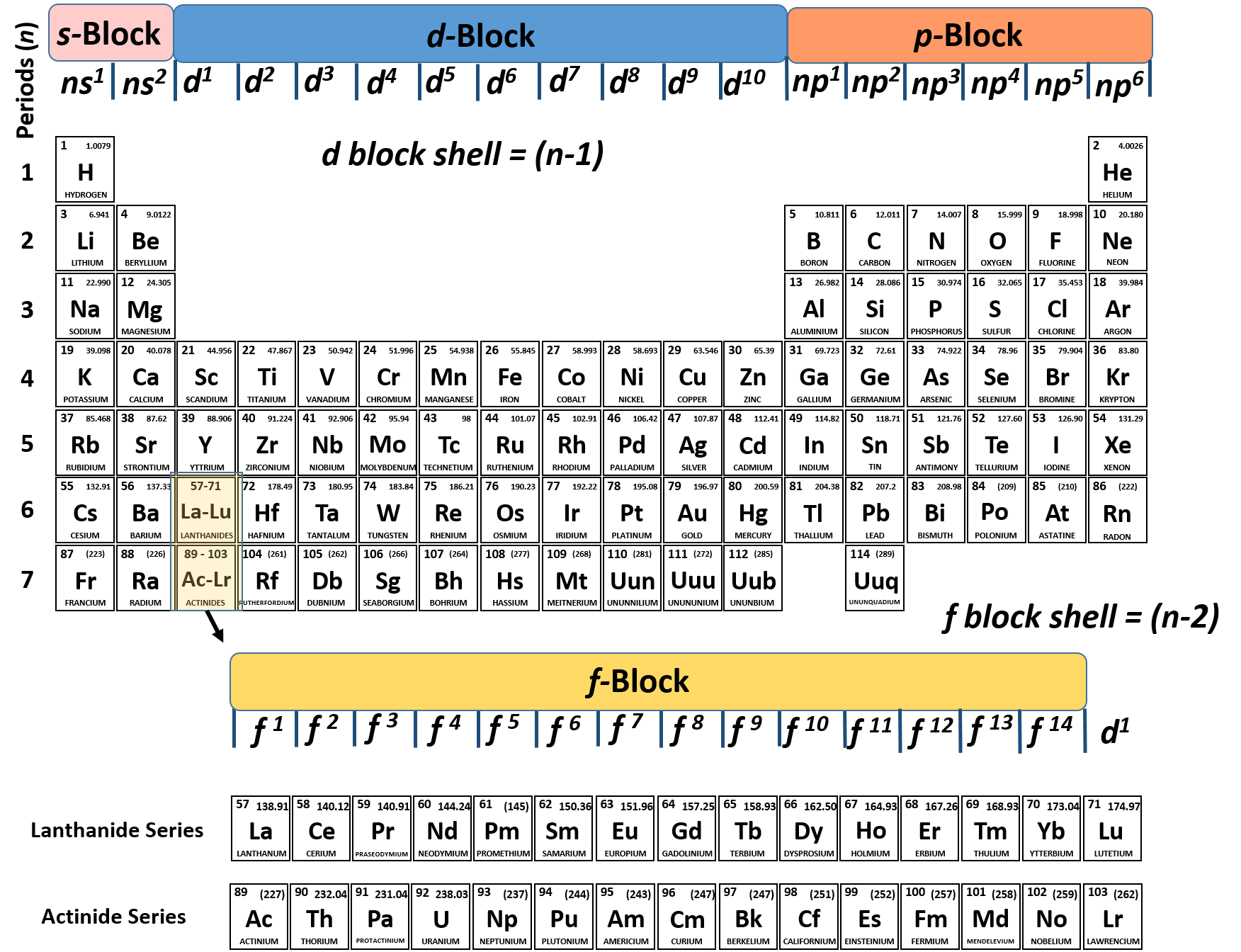
Therefore, electron affinity becomes increasingly negative as we move left to right across the periodic table and decreases as we move down a group. Electron affinity (the energy associated with forming an anion) is more favorable (exothermic) when electrons are placed into lower energy orbitals, closer to the nucleus. Ionization energy (the energy associated with forming a cation) decreases down a group and mostly increases across a period because it is easier to remove an electron from a larger, higher energy orbital. 
Anionic radii are larger than the parent atom, while cationic radii are smaller, because the number of valence electrons has changed while the nuclear charge has remained constant. Covalent radius mostly decreases as we move left to right across a period because the effective nuclear charge experienced by the electrons increases, and the electrons are pulled in tighter to the nucleus. Covalent radius increases as we move down a group because the n level (orbital size) increases. 
View available translations of the content.Įlectron configurations allow us to understand many periodic trends. Download the content in PDF, Microsoft Word, or other format. Questions and Tasks Add a note to the content. Kinetic Molecular Theory, Real GasesĬontent Foreword Atoms Introduction Matter Measurement The Atom Periodic Table Moles & Mass Light Blackbody Radiation, Photoelectric Effect Atomic Spectra, Bohr Model Orbitals & Quantum Numbers Electron Configurations Periodic Trends Molecules Bonding Nomenclature Lewis Structures Part 1 Lewis Structures Part 2 Molecular Shape Polarity Organic Molecules Isomers Valence Bond Theory Molecular Orbital Theory Interactions Pressure & Gas Laws Combined & Ideal Gas Laws Dalton's Law, Graham's Law, Henry's Law Kinetic Molecular Theory, Real Gases Intermolecular Forces Properties of Water Applications of IMF Phase Diagrams Reactions Stoichiometry Limiting Reactants, % Yield % Composition, Empirical Formulas Energy, Heat, and Work Calorimetry Part 1 Calorimetry Part 2 Enthalpy Part 1 Enthalpy Part 2 Entropy Gibb's Free Energy Climate and Human Impacts Search within this publication Search the entire site Blackbody Radiation, Photoelectric Effect 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
